The latest 340B audit news and prevention strategies
Understanding the latest audit activity is crucial for your organization’s 340B compliance success. Included are Kodiak’s audit prevention strategies.
Sept. 9, 2025
Sept. 9, 2025

Staying up to date with evolving regulations and audit findings is crucial for healthcare organizations to remain compliant with 340B Drug Pricing Program requirements. That’s why Kodiak periodically provides you with all the latest updates you need to know (see other recent articles here and here).
The U.S. Department of Health and Human Services’ Health Resources and Services Administration and Office of Pharmacy Affairs are responsible for administering the 340B Program. Since the inception of periodic audits in 2012, HRSA has increased its scrutiny over covered entity compliance with 340B regulations and has consistently audited 200 covered entities each year since 2015.
Since 2012, HRSA has posted more than 2,200 audit results. Below is a summary of recent developments related to HRSA audit findings and HRSA fiscal year 2024 audit results as of August 2025.
HRSA 340B Program integrity audit news
Since 2012, HRSA has communicated its approach for implementing its authority to audit 340B Program stakeholders through policies, guidance documents, and regulations. In 1996, HRSA published “Patient Definition Guidelines,” which contain its interpretation of several key statutory requirements that guide its audit activities. FY 2024 HRSA audit results reflect changes in the enforcement of the “Patient Definition Guidelines.” For example, “340B drugs dispensed at contract pharmacies for prescriptions written at ineligible sites” is listed as the top “Diversion” finding related to FY 2024 HRSA program integrity audits.
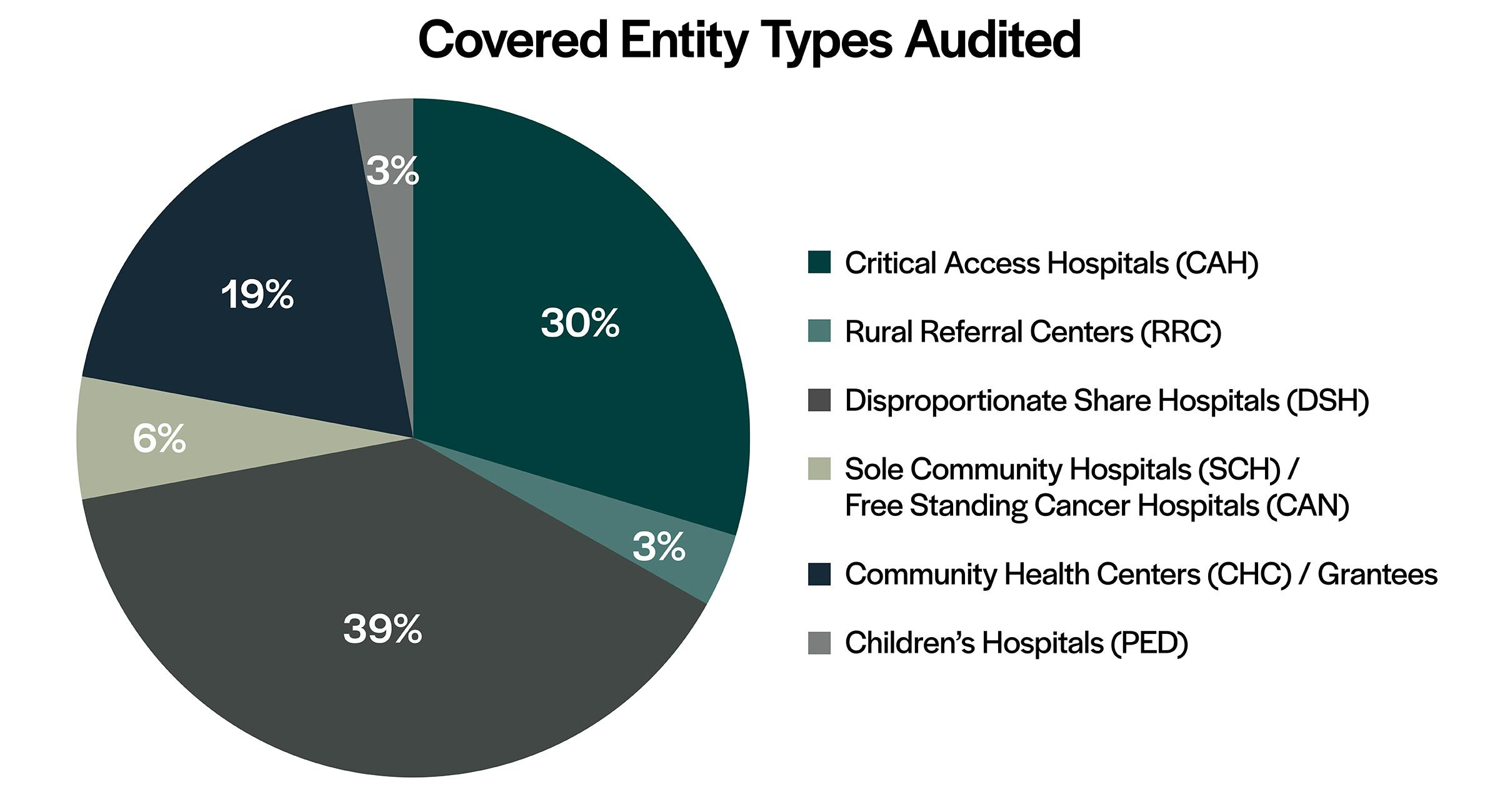
In addition, HRSA continues to be consistent with choosing hospitals and healthcare facilities from a variety of states to audit. Critical access hospitals and disproportionate share hospitals make up most of the audits performed and the total number of findings. Grantees continue to be audited with similar frequency in FY 2024 compared to previous years. In addition, “Incorrect accurate 340B OPAIS Record” remains the top HRSA audit finding in FY 2024, followed by “Duplicate Discount” and “Diversion.”
Following is a summary of FY 2024 HRSA program integrity audit activity
HRSA last updated FY 2024 program integrity audit findings on its website on Aug. 26. The following findings are from the audit results posted to date:
- HRSA posted 172 audit results from FY 2024, with 36% of the audits completed resulting in no adverse findings.
- Hospitals made up 81% of the FY 2024 audits; DSHs made up most of the covered entity types audited (39%), followed by CAHs (30%).
- Grantees, including Community Health Centers, Ryan White HIV/AIDS Program grantees, and Sexually Transmitted Disease Clinics, made up 19% of the covered entities audited in FY 2024.
- HRSA audited covered entities from 44 states.
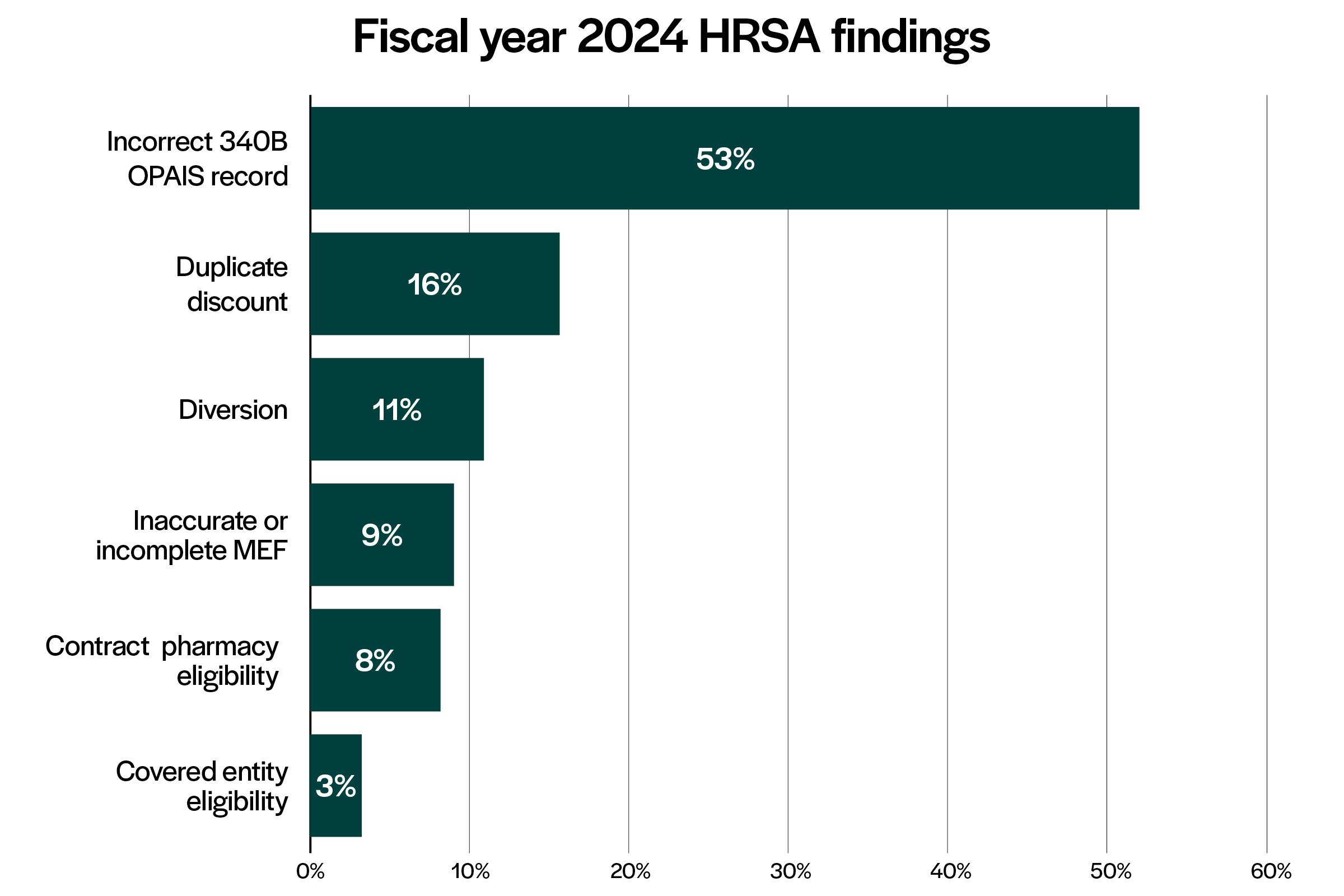
- There are 180 audit findings currently posted for FY 2024 with “Incorrect 340B OPAIS Record” listed as the most common finding (53%), which is consistent with FY 2023 audit findings.
- “Duplicate Discount” is the second most common finding at 16%. “Inaccurate” or “Incomplete Medicaid Exclusion File (MEF)” is listed as the most common of the “Duplicate Discount” findings.
- “Diversion” is the third most common finding at 11%. “340B drugs dispensed at a contract pharmacy for prescriptions written at ineligible sites” is listed as the top finding (70%) in the “Diversion” findings category. It appears that HRSA views contract pharmacy prescriptions written from locations that are not registered as part of the 340B Program to be ineligible to purchase under the 340B Program.
- “Incorrect entry in 340B OPAIS for Medicare Cost Report filing date” is the most common inaccurate 340B OPAIS record finding. “Off-site outpatient facilities were not listed in 340B OPAIS” and “incorrect entry in 340B OPAIS for physical and shipping address” are also common inaccurate 340B OPAIS record findings.
- “Failed to remove duplicate, terminated, or closed contract pharmacies” or “contract pharmacies did not have a written contract in place from the 340B OPAIS” are the most common contract pharmacy eligibility findings.
- An inaccurate or incomplete information on the HRSA MEF finding notes that after further review, a duplicate discount did not occur for the entity that billed Medicaid.
- “Ineligible off-site outpatient facilities registered on 340B OPAIS” is the most common covered entity eligibility finding.
- Of the FY 2024 audits, 24% required repayment to manufacturers due to a “Duplicate Discount” or “Diversion” finding.
- Eight percent of audits require termination of a contract pharmacy location or an off-site outpatient facility from the 340B Program.
- Areas for improvement do not require a corrective action plan.
Audit prevention strategies
Considering these FY 2024 HRSA 340B Program audit findings, organizations should keep the following audit prevent strategies in mind:
- Prevent an “Incorrect 340B OPAIS Record” finding by confirming:
- Contract pharmacy locations registered on the 340B OPAIS match the locations listed in the pharmacy service agreement.
- Closed or terminated contract pharmacies are removed from OPAIS.
- Only eligible sites are registered on OPAIS.
- Duplicate registrations are removed.
- Hospital control type is consistent with eligibility documents.
- The Medicare Cost Report filing date, cost reporting period, and employer identification number on OPAIS are accurate.
- There is accurate physical and ship-to/bill-to address documentation on OPAIS for off-site outpatient facilities, grant-associated sites, contract pharmacies, and entity-owned pharmacies.
- Avoid an “Incorrect 340B OPAIS Record” finding by promptly updating 340B OPAIS with relevant information after submitting a new Medicare Cost Report.
- To help prevent an “Inaccurate MEF” finding, add billing numbers to OPAIS for not only the covered entity’s “home” state but for each state you intend to bill Medicaid fee-for-service for drugs purchased through the 340B Program. In addition, confirm that every child site that chooses to carve-in Medicaid has the appropriate National Provider Identifier and Medicaid Provider Number, and the associated state, listed on the MEF. A complete MEF with accurate billing numbers, NPI, and MPN will also aid in the prevention of a “Duplicate Discount” finding.


